  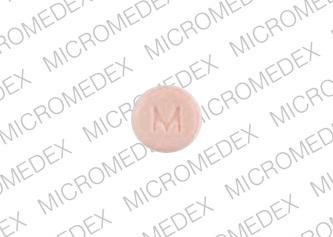
Climara.could Climara be stronger than Vivelle? Hormone therapy just isn't a one size fits all deal, unfortunately. Consumer ratings reports for CLIMARA. Includes 198 patient rankings on scale of 1-5, comments, side effects, dosage, sex, age, time taken. Climara (Estradiol) and/or alternatives. To apply the Climara skin patch, you must place the adhesive side onto a clean and dry section of your lower abdomen or onto the upper area of your buttocks. Climara 5. 0 micrograms/2. Transdermal Patch. For the treatment of postmenopausal symptoms, HRT should only be initiated for symptoms that adversely affect quality of life. In all cases, a careful appraisal of the risks and benefits should be undertaken at least annually and HRT should only be continued as long as the benefit outweighs the risk. Evidence regarding the risks associated with HRT in the treatment of premature menopause is limited. Due to the low level of absolute risk in younger women, however, the balance of benefits and risks for these women may be more favourable than in older women. Medical examination/follow- up. Pictures of Climara (Estradiol Transdermal), drug imprint information, side effects for the patient. Easy to read patient leaflet for Climara weekly patch. Includes indications, proper use, special instructions, precautions, and possible side effects.  
Before initiating or reinstituting HRT, a complete personal and family medical history should be taken. Physical (including pelvic and breast) examination should be guided by this and by the contraindications and warnings for use. During treatment, periodic check- ups are recommended of a frequency and nature adapted to the individual woman. Women should be advised what changes in their breasts should be reported to their doctor or nurse (see 'Breast cancer' below). Investigations, including appropriate imaging tools, e. Conditions which need supervision. If any of the following conditions are present, have occurred previously, and/or have been aggravated during pregnancy or previous hormone treatment, the patient should be closely supervised. It should be taken into account that these conditions may recur or be aggravated during treatment with Climara, in particular: o Leiomyoma (uterine fibroids) or endometriosiso Risk factors for thromboembolic disorders (see below)o Risk factors for oestrogen dependent tumours, e. Hypertensiono Liver disorders (e. The reported increase in endometrial cancer risk among oestrogen- only users varies from 2- to 1. After stopping treatment risk may remain elevated for at least 1. If break- through bleeding or spotting appears after some time on therapy, or continues after treatment has been discontinued, the reason should be investigated, which may include endometrial biopsy to exclude endometrial malignancy. Therefore, the addition of progestogens to oestrogen replacement therapy should be considered in women who have undergone hysterectomy because of endometriosis, if they are known to have residual endometriosis. Breast cancer. The overall evidence suggests an increased risk of breast cancer in women taking combined oestrogen- progestagen and possibly also oestrogen- only HRT, that is dependent on the duration of taking HRT. Combined oestrogen- progestagen therapy. Observational studies have mostly reported a small increase in risk of having breast cancer diagnosed that is substantially lower than that found in users of oestrogen- progestagen combinations (see section 4. The excess risk becomes apparent within a few years of use but returns to baseline within a few (at most five) years after stopping treatment. HRT, especially oestrogen- progestagen combined treatment, increases the density of mammographic images which may adversely affect the radiological detection of breast cancer. Ovarian cancer. Ovarian cancer is much rarer than breast cancer. Long- term (at least 5- 1. HRT products has been associated with a slightly increased risk of ovarian cancer (see section 4. Some studies including the WHI trial suggest that the long- term use of combined HRTs may confer a similar, or slightly smaller, risk (see Section 4. Venous thromboembolism. The size of the Climara and Estradiol patch depends on the strength. The sizes of the Estradiol patches are: 7.75 cm2, 11.625 cm2, 15.5 cm2, 18.6 cm2, 23.25 cm2 and 31 cm2 Climara: 6.5cm2, 9.375 cm2, 12.5 cm2, 15 cm2, 18.75cm2.The occurrence of such an event is more likely in the first year of HRT than later (see Section 4. HRT is therefore contraindicated in these patients (see section 4. There is no consensus about the possible role of varicose veins in VTE. As in all postoperative patients, prophylactic measures need be considered to prevent VTE following surgery. If prolonged immobilisation is to follow elective surgery temporarily stopping HRT 4 to 6 weeks earlier is recommended. Treatment should not be restarted until the woman is completely mobilised. If a thrombophilic defect is identified which segregates with thrombosis in family members or if the defect is 'severe' (e. S, or protein C deficiencies or a combination of defects) HRT is contraindicated. Patients should be told to contact their doctors immediately when they are aware of a potential thromboembolic symptom (e. As the baseline absolute risk of CAD is strongly dependent on age, the number of extra cases of CAD due to oestrogen+progestagen use is very low in healthy women close to menopause, but will rise with more advanced age. Oestrogen- only: Randomised controlled data found no increased risk of CAD in hysterectomised women using oestrogen- only therapy. Ischaemic Stroke. Combined oestrogen- progestagen and oestrogen- only therapy are associated with an up to 1. The relative risk does not change with age or time since menopause. However, as the baseline risk of stroke is strongly age- dependent, the overall risk of stroke in women who use HRT will increase with age (see section 4. Other conditions. Oestrogens increase thyroid binding globulin (TBG), leading to increased circulating total thyroid hormone, as measured by protein- bound iodine (PBI), T4 levels (by column or by radio- immunoassay) or T3 levels (by radio- immunoassay). T3 resin uptake is decreased, reflecting the elevated TBG. Free T4 and free T3 concentrations are unaltered. Other binding proteins may be elevated in serum, i. Free or biological active hormone concentrations are unchanged. Other plasma proteins may be increased (angiotensinogen/renin substrate, alpha- I- antitrypsin, ceruloplasmin). Women with a tendency to chloasma should minimise exposure to the sun or ultraviolet radiation whilst taking HRT. There is some evidence of increased risk of probable dementia in women who start using continuous combined or oestrogen- only HRT after the age of 6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |
 RSS Feed
RSS Feed
